원문링크: https://www.nature.com/articles/s41467-024-46909-1
Abstract
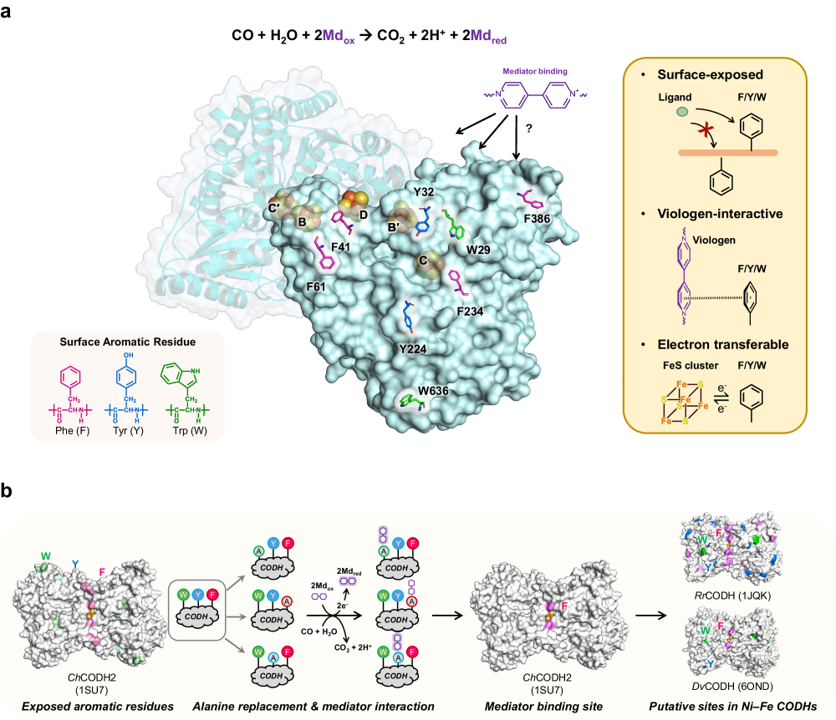
Fe‒S cluster-harboring enzymes, such as carbon monoxide dehydrogenases (CODH), employ sophisticated artificial electron mediators like viologens to serve as potent biocatalysts capable of cleaning-up industrial off-gases at stunning reaction rates. Unraveling the interplay between these enzymes and their associated mediators is essential for improving the efficiency of CODHs. Here we show the electron mediator-interaction site on ChCODHs (Ch, Carboxydothermus hydrogenoformans) using a systematic approach that leverages the viologen-reactive characteristics of superficial aromatic residues. By enhancing mediator-interaction (R57G/N59L) near the D-cluster, the strategically tailored variants exhibit a ten-fold increase in ethyl viologen affinity relative to the wild-type without sacrificing the turn-over rate (kcat). Viologen-complexed structures reveal the pivotal positions of surface phenylalanine residues, serving as external conduits for the D-cluster to/from viologen. One variant (R57G/N59L/A559W) can treat a broad spectrum of waste gases (from steel-process and plastic-gasification) containing O2. Decoding mediator interactions will facilitate the development of industrially high-efficient biocatalysts encompassing gas-utilizing enzymes.
